In Practice
Follow this topic
Bookmark
Record learning outcomes

"A number of pharmacies" are making incorrectly endorsed claims for medicines dispensed through the Government’s new serious shortage protocols, PSNC warned today (October 14).
PSNC said that since the introduction on October 3 of the first SSPs for certain strengths of fluoxetine capsules, it has been working with the NHS Business Services Authority “to understand if claims are being submitted in accordance with SSP endorsement guidance”.
NHSBSA told PSNC it had received “a number of EPS messages which do not clearly indicate the relevant endorsed information required for a valid SSP claim,” the negotiator reported.
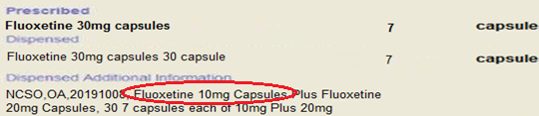
In one example highlighted by NHSBSA (see below image) a contractor attempting to claim via SSP02 – which allows pharmacists to substitute a different formulation to fluoxetine 30mg capsules – specified 10mg capsules, which are currently affected by shortages, rather than 10mg tablets as authorised by the SSP.
PSNC told PNN that as the fluoxetine capsules currently hit by shortages are "fairly low volume lines" it did not expect to see large numbers of SSP endorsements, adding that because October prescriptions may be submitted until midnight on November 5 "we cannot yet ascertain how common these inaccurate SSP claims are".
PSNC advised that pharmacies double check that SSP endorsements reflect the requirements outlined in the corresponding SSP, and include ‘NCSO’ endorsement.
Contractors must also “ensure the correct quantity of the alternative is endorsed to cover the equivalent number of days’ treatment as indicated on the original prescription,” the negotiator emphasised.
The fluoxetine SSPs are due to expire on October 31, although according to PSNC this date “may be brought forward or extended, and the content may be amended throughout this period”.
Contractors who have made incorrect or incomplete SSP endorsements have until the same date to rectify their claims using the EPS claim amend facility.
The full service specifications for the fluoxetine SSPs can be found on the NHSBSA website.

